The curious case of MTHFR gene diagnoses
You may have heard people talk about the MTHFR gene as if it holds the key to all sorts of health problems. In clinical practice, however, it’s a very different story. During my years working in acute hospital care, I saw only a handful of genuine MTHFR (methylenetetrahydrofolate reductase)-related cases — and there’s a good reason for that.
When Is MTHFR Testing Actually Needed?
According to UK and international guidelines, MTHFR testing is only recommended in very specific situations. The most common is in infants or children with signs of a severe metabolic disorder, particularly when dangerously high homocysteine levels cause neurological symptoms. This condition, called homocystinuria, is rare and serious.
Testing may also be considered when someone — or their close relatives — has a history of early heart disease, stroke, or unexplained blood clots, especially if homocysteine levels are raised. In some cases, it may be explored alongside other investigations for recurrent pregnancy loss or pre-eclampsia.
Outside of these scenarios, routine MTHFR testing simply isn’t clinically useful.
The Rise of Private MTHFR Testing
Despite this, more and more people are turning up with private test results showing “MTHFR deficiency”, often recommended by functional medicine or non-mainstream practitioners. This trend isn’t supported by evidence, and it isn’t reflected in any clinical guideline worldwide.
What We Actually Know About MTHFR
Interest in MTHFR began in the 1990s, when researchers were exploring homocysteine as a potential cardiovascular risk factor. They already knew that children with homocystinuria — often caused by mutations in a different gene — developed severe, early-onset vascular disease.
Today, we know that MTHFR variants are extremely common. Around 60–70% of the global population carries one. They are most prevalent in Hispanic populations, followed by white populations, especially those from Mediterranean regions.
But here’s the key point: having a variant does not mean you have a health problem (Michot et al., 2008), and it is very different from methylenetetrahydrofolate reductase (MTHFR) deficiency, which is a rare autosomal‑recessive inherited disorder. About 100 patients have been reported since its first description in 1972 (Michot et al., 2008)
Most people with one — or even two — of the common variants process folate normally and never experience symptoms. These variants are considered part of normal human genetic diversity. This is also why countries such as Australia and now the UK fortify flour with folic acid: it ensures adequate folate intake for everyone, regardless of genetics. Most European countries, including Poland and Italy, do not encourage the food industry to do so, and that is based on concerns raised by those on the decision-making panel about masking vitamin B12 deficiencies, among other issues. I feel it is a weak reason, because those concerns mainly relate to the older population, but it begs a question: what about the younger population, children, and females of reproductive age?
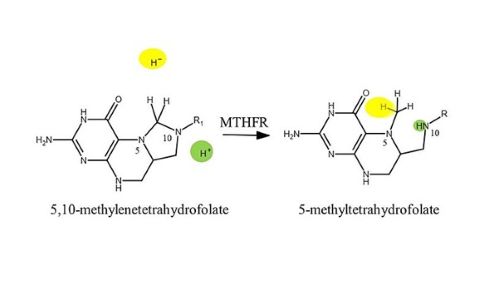
What Does MTHFR Actually Do?
source: scienedirect.
MTHFR genes instruct the body on how to produce the MTHFR enzyme, which is directly involved in converting digested folate into the active form of vitamin B9 (5‑methyltetrahydrofolate) needed for the remethylation of homocysteine to methionine. If you have an MTHFR gene variant, your body may not process folate as efficiently as it should, which can impair the methylation pathway and, in turn, hinder the body’s ability to process histamine. In a very small number of people, this can lead to raised levels of homocysteine, an amino acid that may contribute to health problems. High homocysteine has been linked to vascular and neurological risks. However, for the vast majority of people, MTHFR variants do not cause this issue.
The Bottom Line
It is true that when MTHFR function is reduced, your nutrient requirements may increase — particularly for folate, vitamins B12, B2 and B6, as well as choline and magnesium (Serapinas, et al, 2017). Most people with MTHFR variants, however, are completely healthy.
If you’ve had a private test and discovered a variant, it does not automatically explain symptoms or health concerns. A far more useful — and clinically relevant — step is to check your folate levels. If they are low, improving your diet or using appropriate supplementation is usually all that is required.
Put simply, for most people, folate status is far more important than MTHFR status — and that is what the scientific evidence shows
Why is Vitamin B9 Important for Our Health?
Vitamin B9 is essential for health throughout life. From the very beginning of pregnancy, it plays a crucial role in supporting the healthy development of the foetus, particularly in early pregnancy, when it helps reduce the risk of birth defects affecting the brain and spine. Folate is also vital for DNA and RNA formation and repair, ensuring that cells receive the correct instructions to divide, repair themselves, and form new tissues when needed. This makes it fundamental for the growth and maintenance of all tissues, including those required for healthy skin, hair, and nails.
Folate is also important for the health of blood vessels and the intestinal tract, and it is involved in the production of red blood cells, helping reduce the risk of anaemia. Although medical researchers are not entirely certain whether raised homocysteine levels directly cause health problems, some studies suggest that high homocysteine may damage the lining of blood vessels. This may increase the risk of blood clots, heart disease, and stroke.
In short, Vitamin B9 (folate) is important for:
- methylation (involved in over 200 biochemical reactions)
- detoxification
- DNA repair
- neurotransmitter balance
- healthy pregnancy and fertility
- energy production
- homocysteine regulation
Beyond folate absorption, the MTHFR gene is heavily involved in the methylation process, which converts homocysteine into methionine — a substance required for normal metabolism, muscle growth, and the production of glutathione.
How Much Folate Do You Need?
Requirements vary, but UK recommendations from the British Dietetic Association include:
- Adults and children over 11: 200 μg dietary folate per day
- Women planning pregnancy: a 400 μg folic acid supplement daily until the 12th week of pregnancy, alongside a 300 μg daily dietary intake, to reduce the risk of neural tube defects
- High‑risk cases: may require a 5 mg supplement, as advised by a clinician
People with certain medical conditions — including coeliac disease, where absorption may be impaired — may be advised to consume higher amounts. This is assessed individually and may also apply to individuals living with obesity or diabetes, or those taking anti‑epileptic medication.
MTHFR, Perimenopause, and Symptoms
Clinical evidence suggests that signs of MTHFR (methylenetetrahydrofolate reductase) dysfunction may become more noticeable or worsen during perimenopause. This is due to declining oestrogen levels — which normally help lower homocysteine — combined with reduced methylation efficiency (the production of methyl groups). This metabolic shift may contribute to intensified mood disorders, fatigue, cognitive decline (“brain fog”), and increased cardiovascular risk (Andrade et al., 2025).
What If You Have Had an MTHFR Test and Found a Variant?
If you have paid for private MTHFR testing and discovered a variant, it is sensible to inform your GP.
From a dietetic perspective, the focus remains the same: eat a well‑balanced diet rich in vitamins B6, B12 and folate (B9), and remember that magnesium and choline are also important. For perimenopausal and menopausal women — or in any situation where female hormonal imbalance occurs — it is also beneficial to include phytoestrogen‑rich foods.
No nutrient works in isolation. Folate requires adequate amounts of other B‑group vitamins, magnesium, and choline to function effectively. Folate and vitamin B12 work together to form healthy red blood cells, and iron is also essential for producing them. Folate and B12 also support the nervous system, alongside magnesium and choline. Adequate intake of these nutrients helps prevent folate‑dependent deficiencies.
Lifestyle factors that may reduce folate metabolism include smoking, alcohol consumption, and high intakes of coffee. If you are planning a pregnancy, follow the usual recommendation of 0.4 mg/day of folic acid (or a higher dose if advised by a clinician).
UK and international guidelines emphasise addressing diet and lifestyle first, unless a clinically complex situation has been identified — which, as noted earlier, affects only a very small percentage of the global population.
Dietary Sources of Folate
If you are deficient in folate — whether or not you know your MTHFR status — diet is essential in helping you meet your needs. Folate cannot be stored in the body, so it must be consumed daily. A varied diet that includes all major food groups is the most reliable way to ensure adequate intake.
Rich dietary sources include:
- spinach, kale, Brussels sprouts, cabbage, broccoli
- beans and legumes (chickpeas, lentils, black‑eyed beans, kidney beans)
- nuts and seeds
- oranges and orange juice
- wheat bran and wholegrain foods (more challenging for those on a gluten‑free diet)
- poultry, pork, shellfish, liver
- yeast extracts such as Marmite
- beef extracts
- eggs — also an excellent source of choline, vitamin B12, vitamin B2, iron, vitamin D, and selenium
Fortified foods (e.g., some breakfast cereals) can also contribute — always check the label.
Because folate is water‑soluble, a significant amount may be lost during cooking unless the cooking water is consumed (e.g., in soups or stews). Alcohol, smoking, and heavy consumption of tea or coffee may also reduce folate levels (Otake et al., 2018). Some medications, such as diuretics, may have an impact as well.
The Take‑Home Message
Knowing whether you have an MTHFR variant often does not change clinical management. For example, in pregnancy, it is more effective — according to Long & Goldblatt (2016) — to measure maternal serum folate levels and supplement accordingly, rather than test for MTHFR polymorphisms. Measuring homocysteine levels may also provide useful information about neural tube defect risk.
Regardless of MTHFR status, 400 μg of folate daily is recommended for women of reproductive age. A couple’s MTHFR polymorphism status does not alter pregnancy‑related management for preventing primary or recurrent neural tube defects.
Researchers and clinicians also highlight that routine MTHFR testing does not benefit the general population and may contribute to unnecessary health anxiety.
The key message is this:
unless wider complications are identified (such as significantly raised homocysteine), the priority is to check for nutritional deficiencies — particularly folate — and address them through diet or supplementation where needed.
Data from countries such as Australia show that where folate supplementation is routine, MTHFR mutations do not significantly raise homocysteine levels — a risk factor for cardiovascular disease, cognitive decline, and reduced bone density, among others (Deloughery et al., 2022).